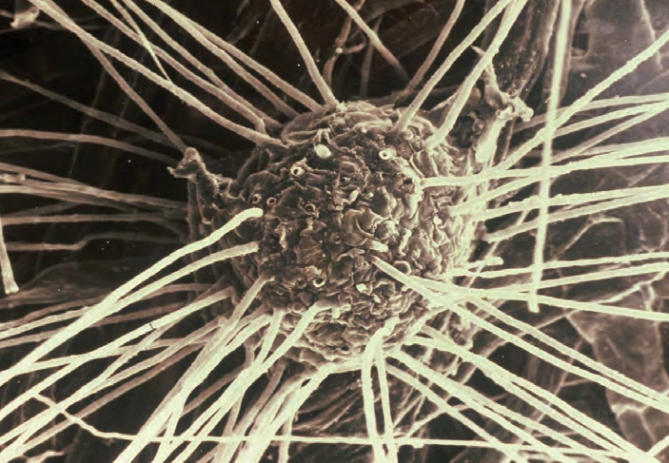
Let’s begin with a topic that interests all farmers and one to which nearly all the other benefits of mycorrhizae are inherently linked: improving crop yields. Typically, mycorrhizae’s single most prominent contribution to a crop plant is improved access to and uptake of phosphorus (P).
All farmers are intensely familiar with the importance of this elemental nutrient to essential plant functions, which include energy transfer, photosynthesis, the transformation of carbohydrates, systemic nutrient mobilization, and genetic transfers. Given that often one of the most noticeable evidence of P deficiency in a crop is reduced yield (or in forage and pasture reduced quantity), it is no wonder that P is such a critical (and expensive) component in crop fertilizers.
Much of the naturally-occurring P in soils is found bound tightly with elements such as iron or aluminum in the form of recalcitrant compounds. Similarly, P inputs derived from fertilizers often react with ambient soil cations to form insoluble salts. In natural ecosystems, plant communities rely on mycorrhizal fungi to access these forms of phosphorus.
Mycorrhizal hyphae produce enzymes, including phosphatase to convert phosphorus into soluble, plant-usable forms. This same process can be valuable in agriculture, maximizing the availability of natural soil P as well as dramatically enhancing the efficient uptake of P derived from fertilizers. With greater P uptake, costs go down and yields frequently increase as well.
The availability of nitrogen can also be a factor in limiting crop productivity for reasons opposite to those limiting P. Available nitrogen in the forms of nitrates (N03), nitrites (N02), and ammonium (NH4) are very soluble and can flow past the root zone before roots can absorb it. This means they are often lost to run-off or groundwater or trapped in subsoil beyond the access of roots.
The profoundly dense network of tiny hyphae filaments in a mycorrhizal system typically extends 45 to 60 centimeters beyond the roots themselves, increasing the absorptive surface area of colonized roots hundreds to thousands of times. A teaspoon of mycorrhizal soil can easily contain several kilometers of hyphae, all of which are highly absorptive of soluble nitrogen ions, ensuring optimum uptake and the associated nitrogen-related cropping benefits.
Another source of nitrogen uptake unique to mycorrhizal symbiosis has recently been discovered by scientists at the University of California, Irvine, US. The researchers set out to explore how nutrients, including nitrogen, are mobilized through the environment. Using cutting-edge technology, nanometre-sized bits of a semiconducting material called quantum dots were attached to organic compounds such as nitrogen-laden amino acids.
Nutrient transfer discovery
When energised by an ultraviolet laser, the tiny dots emitted light, becoming detectable by special cameras positioned in the root zone of plants. In this manner, the scientists could track nutrients as they were absorbed into the microscopic mycorrhizal hyphae and follow their subsequent movement into the tissue of the host plant.
For more than 100 years conventional scientific wisdom held that root absorption of nitrogen was restricted to inorganic forms of nitrogen such as N03, N02, and NH4. But to their surprise, the scientists saw the illuminated dots attached to amino acids enter the mycorrhizal hyphae and observed them as entire molecules moved into the root cell vacuoles and then continued systemically to the chloroplasts (in which nitrogen is used for photosynthesis).
MYCORRHIZAE

In non-mycorrhizal rhizospheres, amino acids, which are the primary components of proteins, must undergo extensive and time-consuming decomposition processes by bacteria and other soil organisms before nitrogen is released in inorganic, plant-usable forms. In many cases, much of the nitrogen is consumed by the organisms, further delaying its plant availability.
This research demonstrates that mycorrhizal fungi allow their plant hosts to bypass this process, implementing quick and effective access to organic nitrogen sources. What this means to the farmer is that utilizing mycorrhizal fungi, naturally occurring and introduced sources of organic nitrogen (such as found in fish-based fertilizers, green manures, and compost) can provide a readily available source of nitrogen to promote crop growth and enhance yields.
In addition to phosphorus and nitrogen, the mass of hyphal filaments in the soil surrounding mycorrhizae-colonized roots is also capable of mobilizing an array of other important plant nutrients, including calcium, iron, magnesium, and critical micro-nutrients such as manganese, zinc, and copper. Just as a lack of vitamins can impair human or animal health, crop yields and forage production are sometimes limited by insufficient supplies of these minor- and micro-nutrients, even when N-P-K is abundant.
Mycorrhizae’s ubiquitous presence throughout the surrounding soil can access these relatively scarce resources and, in many cases, can release them from insoluble compounds via the production of specialized enzymes. The management of micro-nutrients is becoming increasingly recognized as an important component of modern cropping science. Mycorrhizal fungi can serve as a useful tool to ensure that both natural and introduced sources of these nutrients are transferred efficiently from the soil to the plant.

Help find water
Mycorrhizae’s significant assistance with nutrient uptake is important, but it is not the only crop-enhancing benefit offered by these amazing fungi. Another valuable feature is water management. The expanded and enormous absorptive surface area connected to the roots is going to ensure that nearly all moisture in a plant’s surrounding soil is accessed. But what then? Once the soil is dry, how can the plant survive?
Mycorrhizae provide a mechanism inside the root cells that addresses this problem. When a root cell becomes colonized by a mycorrhizal fungus, a special shared organ called a vesicle grows inside the root cell. The vesicle is essentially a storage container for water and dissolved nutrients that can be utilized in times of deficiencies, such as drought periods.
When moisture and nutrients are abundant in the soil, surplus supplies are stored in the vesicle. When moisture and/or nutrient shortages occur, the plant begins to utilize the resources stored in the vesicles to avoid stress for extended periods – often weeks or even months longer than non-mycorrhizal plants.
When moisture or nutrients again become available, the plant is able to return to normal, healthy respiration and growth without shock or other negative symptoms. Of course, the reservoir provided by the vesicle cannot last indefinitely and the plant will suffer stress and ultimately death if sufficient moisture or nutrients remain unavailable for too long.
However, in most cases the extra non-stressed time provided via the vesicle allows the plant to survive until the next rainfall. This is great news for the dryland farmer. Australia’s recent excessive rainfall notwithstanding, drought is a serious risk encountered by all dryland farmers. Although not infallible, mycorrhiza inoculation offers inexpensive crop insurance as one of its many benefits.